ASTM F3631: Standard Test Method for Assessment of Intra-operative Durability of Intervertebral Body Fusion Devices
ASTM F3631 establishes standardized procedures for evaluating the impact durability of lumbar intervertebral body fusion devices (IBFDs) during surgical insertion. This test method aims to simulate the intra-operative conditions to ensure that IBFD assemblies, comprising the implant and its associated inserter tool, can withstand the mechanical stresses encountered during implantation.
ASTM F3631 outlines two primary test methods:
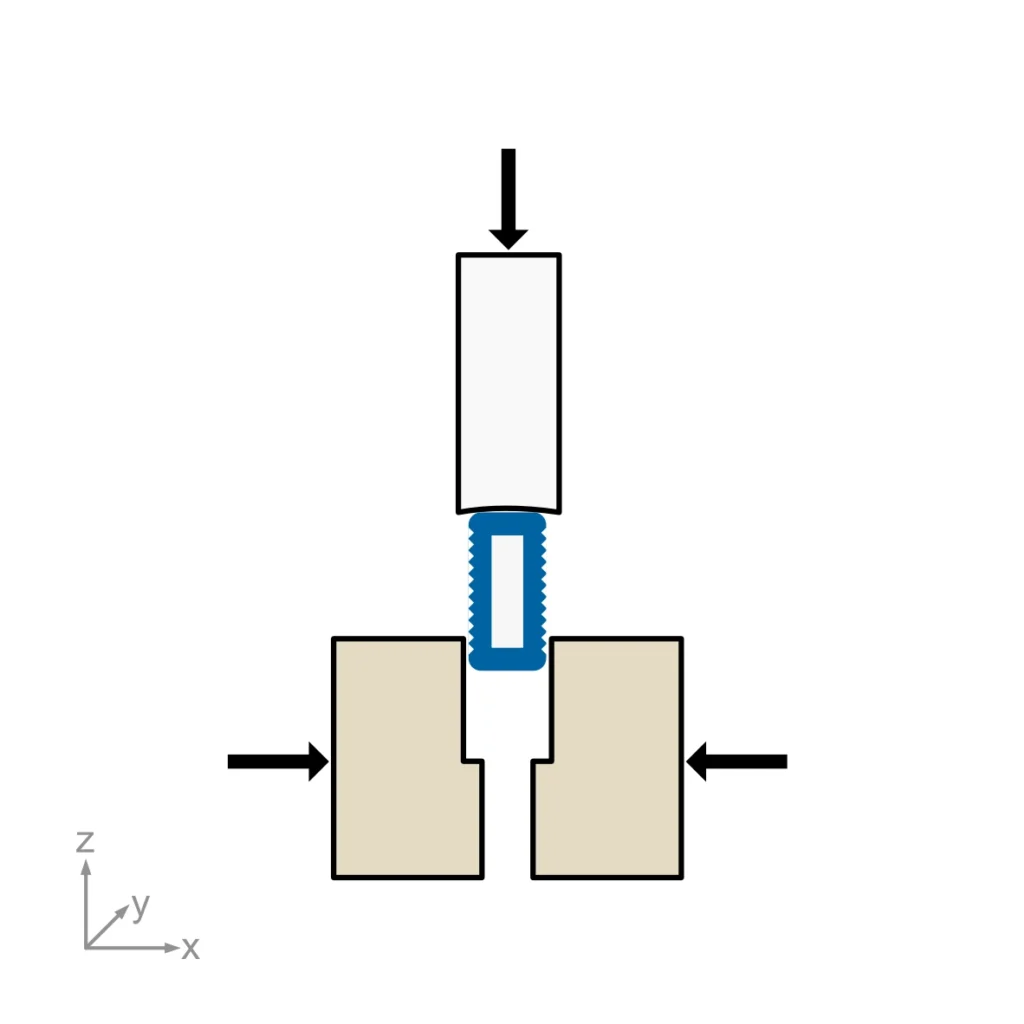
- Insertion Between Foam Blocks: This method simulates the insertion phase by using two Grade 40 polyurethane foam blocks to represent adjacent vertebral bodies. A constant axial preload is applied across the IBFD/foam block assembly, and impact loads are delivered using a drop weight apparatus. A hard stop (“lip”) is incorporated into the foam blocks to allow for continued impact loading until failure or completion of a 40-impact test regimen.
- Static Rigid Block: This method evaluates the impact resistance of the IBFD assembly against a stainless steel block. The IBFD and inserter instrument are mounted in a test apparatus, and impact loads are applied to assess the device’s durability under simulated surgical conditions.
Both methods include options for stair-step loading schemes to assess the impact resistance of the IBFD assemblies systematically.
At Endolab, we have developed a proprietary test setup for horizontal load application and dynamic measurement, integrated into our advanced drop tower system. This cutting-edge technology allows us to seamlessly adapt to our clients’ implant designs and test protocols, ensuring precise and reliable results tailored to your specific needs.
Discover how Endolab’s expertise can support your implant testing requirements with accuracy and efficiency.