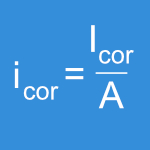ASTM F2003: Standard Practice for Accelerated Aging of Ultra-High Molecular Weight Polyethylene After Gamma Irradiation in Air
ISO 5834-3: Implants for surgery – Ultra-high-molecular-weight polyethylene – accelerated ageing methods
This practice describes a laboratory procedure for accelerated aging of ultra-high molecular weight polyethylene (UHMWPE) specimens and components for total joint prostheses. The UHMWPE is aged at elevated temperature and at elevated oxygen pressure, accelerating the material’s oxidation and thereby allowing for the evaluation of its long-term chemical, physical and mechanical stability.
The accelerated aging method described in this practice allows the investigator to compare the oxidative stability of different UHMWPE materials. However, it is recognized that this method may not precisely simulate the degradative mechanisms of an implant during real-time shelf aging and implantation.