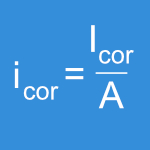ASTM F2129: Standard Test Method for Conducting Cyclic Potentiodynamic Polarization Measurements to Determine the Corrosion Susceptibility of Small Implant Devices
This test method evaluates the corrosion susceptibility of small medical implant devices, single device components or other material samples made of metal. The test is performed using potentiodynamic polarization in a cyclic manner (forward and reverse). For measurement, a high-precision computerized potentiostat is used.
Small medical implant devices covered by this standard include but are not limited to, stents (vascular and ureteral), filters, grafts, occluders, staples and aneurysm or ligation clips. Phosphate buffered saline (PBS) is used as the standard test fluid, but other simulated physiological solutions are also possible.