ASTM F2789: Standard guide for Mechanical and Functional Characterization of Nucleus Devices
This guide describes various forms of nucleus replacement and nucleus augmentation devices. It further outlines the types of testing that are recommended in evaluating the performance of these devices.
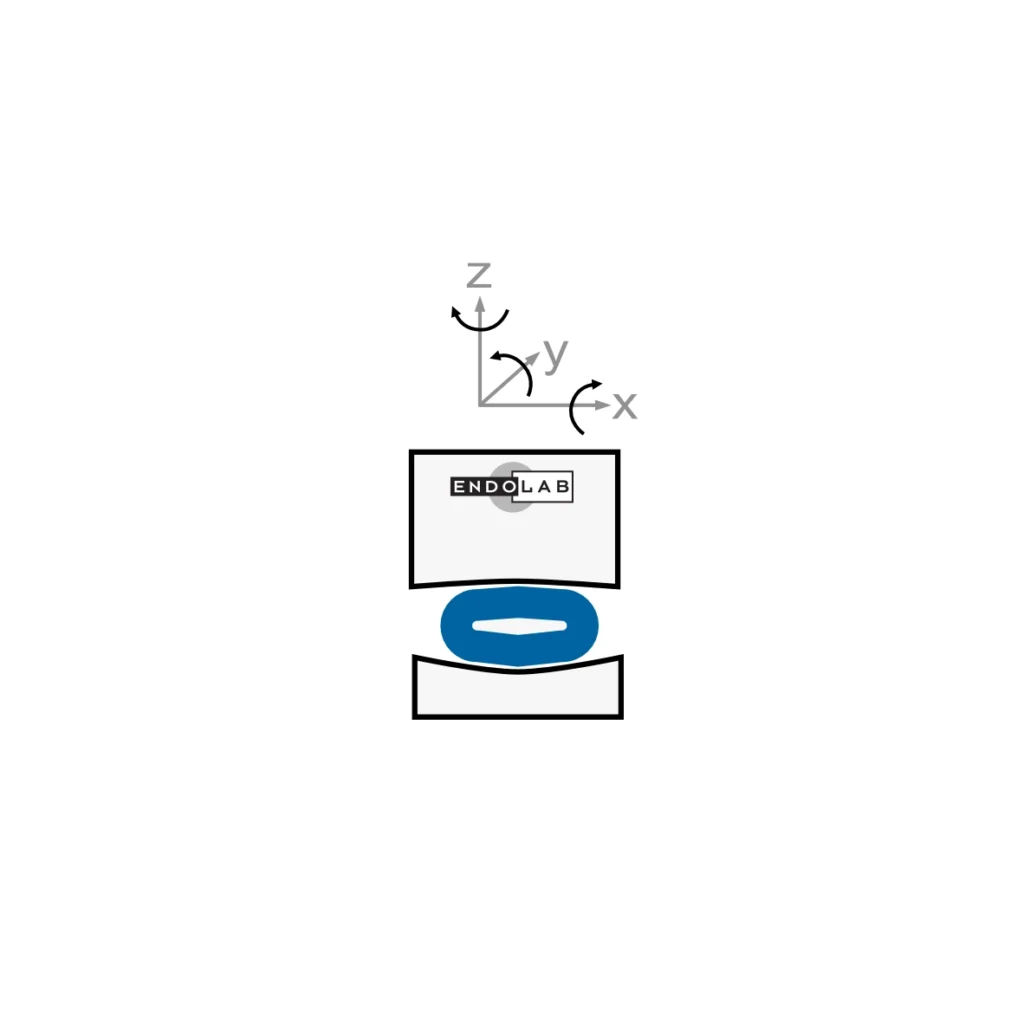
While ISO 18192-2 (wear testing) avoids the use of an annulus replicate, ASTM F2789 describes the manufacturing method and material of such a device. Additional tests such as creep, viscoelasticity, aging and swelling are defined. Endolab® does perform the complete set of tests given by the test standard.