ASTM F2267: Standard Test Method for Measuring Load Induced Subsidence of Intervertebral Body Fusion Device Under Static Axial Compression
This test method specifies the materials and methods for the axial compressive subsidence testing of non-biologic intervertebral body fusion devices, spinal implants designed to promote arthrodesis at a given spinal motion segment.
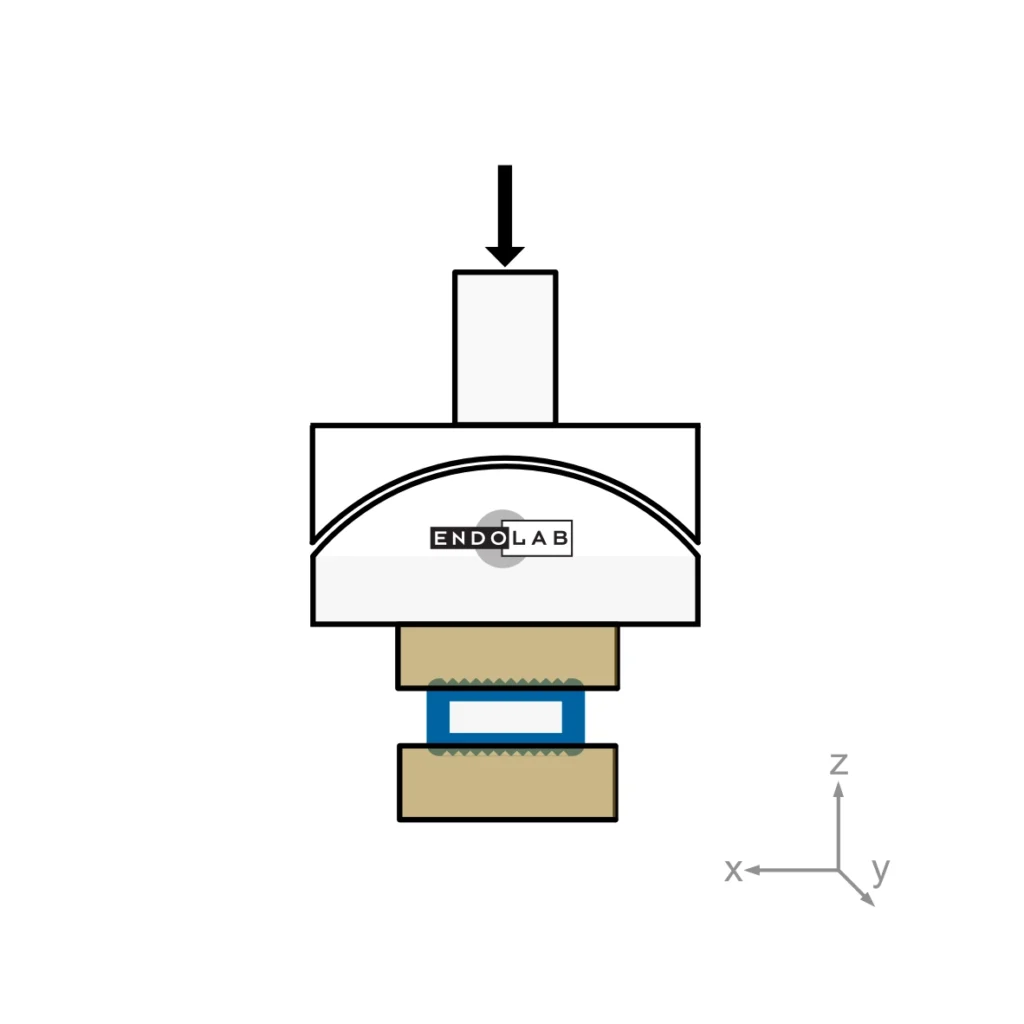
ASTM F2267 is part of the tests required by the FDA for cages. The cage is placed between simulated bone material and an axial load is applied. The amount of deflection is recorded and corrected by the stiffness of the test set-up. The results can be compared to the EndoLab® database.