ISO 25539‐1: Cardiovascular implants – Endovascular devices – Part 1: Endovascular prostheses.
ISO 25539‐2: Cardiovascular implants – Endovascular devices – Part 2: Vascular stents.
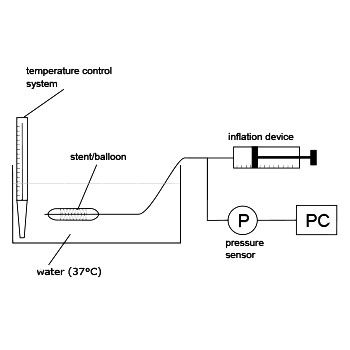
The purpose of this test is to evaluate the ability of the balloon to withstand repeated inflation cycles. Using clinically relevant rates, the balloon is inflated to the RBP or maximum diameter for a preset time and deflated again. These steps are repeated for a minimum of 10 cycles.