ASTM F732: Standard Test Method for Wear Testing of Polymeric Materials Used in Total Joint Prostheses
This test method describes a laboratory method for evaluating the wear properties of polymeric materials intended for use as bearing surfaces in human total joint prostheses. The standard utilizes simplified specimen geometries to rank material combinations based on their wear rates under simulated physiological conditions.
EXECUTIVE SUMMARY
ASTM F732 is a standardized laboratory test designed to determine the wear rate of polymers used in medical implants. It measures the material loss from a polymeric component—such as UHMWPE or PEEK—when articulating against a metallic or ceramic counterface under simulated body conditions. This test serves as an essential first screening step, offering a simpler and more cost-effective alternative to full joint simulators. It allows developers to create a performance ranking of material combinations, ensuring that only the most promising candidates advance to more complex and expensive evaluations.
IN-DETAIL TEST METHOD EXPLANATION
Significance and Methodology
The primary objective of the test is the comparison of new material formulations against established clinical standards. By isolating the material interaction from complex implant geometries, the test identifies how specific variables—such as cross-linking density or sterilization methods—influence wear behavior.
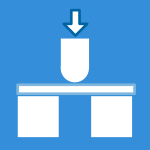
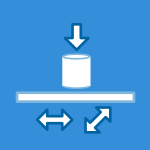
- The Test Apparatus: Testing is typically performed on “Pin-on-Flat” or “Pin-on-Disk” machines. This simplified geometry uses a small pin rubbing against a flat plate, representing the fundamental mechanical interaction of a joint.
- The Result: The primary measurement is the mass loss of the polymer, which is converted into volume loss (mm3). Reporting in volume is necessary to accurately compare materials with different densities.
- Standard Comparison: To ensure validity, new materials are tested alongside a reference standard, typically conventional UHMWPE articulating against a cobalt-chromium alloy.
Controlled Test Parameters
To achieve realistic and reproducible results, laboratory conditions must be strictly controlled to mimic the environment of the human body:
- Fluid (Lubricant): Bovine blood serum is used as the lubricant to simulate synovial fluid. The protein concentration is typically maintained at 20–30 g/L. To prevent calcium phosphate precipitation, which can act as an abrasive, EDTA is added to the solution.
- Temperature: The test system is precisely maintained at body temperature (37±3∘C).
- Duration: Tests generally run for at least two million cycles to gather statistically significant long-term wear data and to move past the initial “run-in” phase of the material.
Kinematics and specialized Annexes
The standard distinguishes between different types of motion, as the wear mechanism in a hip joint differs fundamentally from that in a knee joint or a finger joint:
- Annex A1 – Linear Motion: For applications where materials primarily slide back and forth, such as in simple hinge joints.
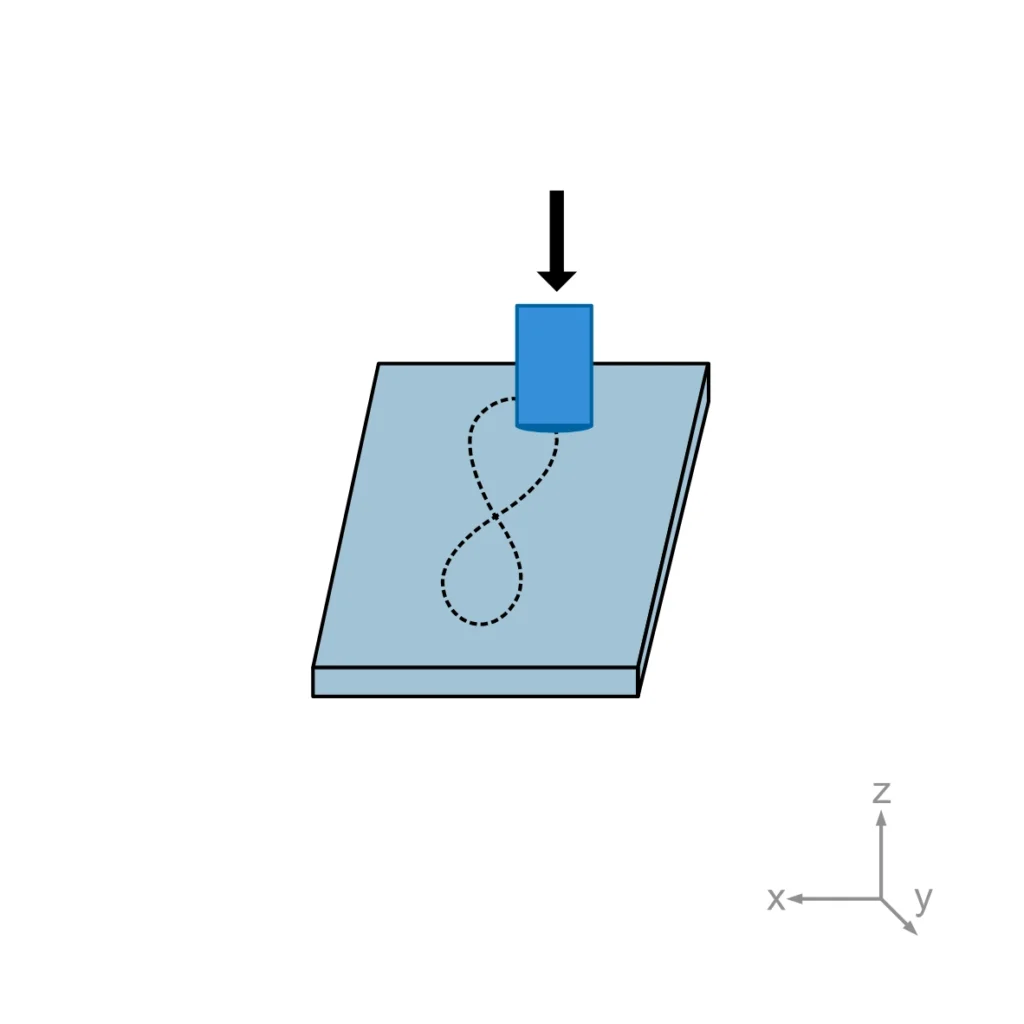
- Annex A2 – Ball-Cup System: Specifically for the biaxial motion of hip joints. This annex introduces “cross-shear,” where the sliding direction changes relative to the polymer’s molecular orientation, which is a known accelerator of wear in vivo.
- Annex A3 – Delamination: A specialized test to determine if a material is prone to surface damage, such as flaking or sub-surface cracking, which is a common failure mode in high-stress knee applications.
Accuracy through Soak Controls
A critical technical challenge in polymer testing is fluid sorption. Because plastics can absorb water and proteins from the lubricant, they often gain weight during the test. If this weight gain is not accounted for, the measurement of material loss would be incorrectly low.
To ensure accuracy, EndoLab utilizes “control soak specimens.” These are identical polymer pieces placed in the same fluid and subjected to the same loads but without the rubbing motion. The weight gain of these controls is subtracted from the results of the active test samples, isolating the true material loss due to wear.
Manufacturing and Sterilization
Specimens must be representative of the finished medical device. The standard requires that test samples undergo the same manufacturing processes—such as compression molding or machining—and the same sterilization cycles (e.g., Gamma irradiation or Ethylene Oxide) as the final commercial implant. Sterilization is known to alter the cross-linking and oxidation state of polymers, which directly impacts wear resistance.
Final Documentation and Analysis
The final test report provides more than just a numerical wear rate. It includes a detailed characterization of the worn surfaces using high-resolution imaging, such as Scanning Electron Microscopy (SEM). This documentation confirms that the wear patterns, such as polishing or scratching, resemble those expected in clinical retrievals.
EndoLab’s expertise in ASTM F732 ensures that manufacturers receive a robust, audit-ready data package. This data is essential for the “Performance Testing – Bench” section of regulatory submissions, providing the empirical evidence needed to confirm the safety and longevity of new polymeric biomaterials.