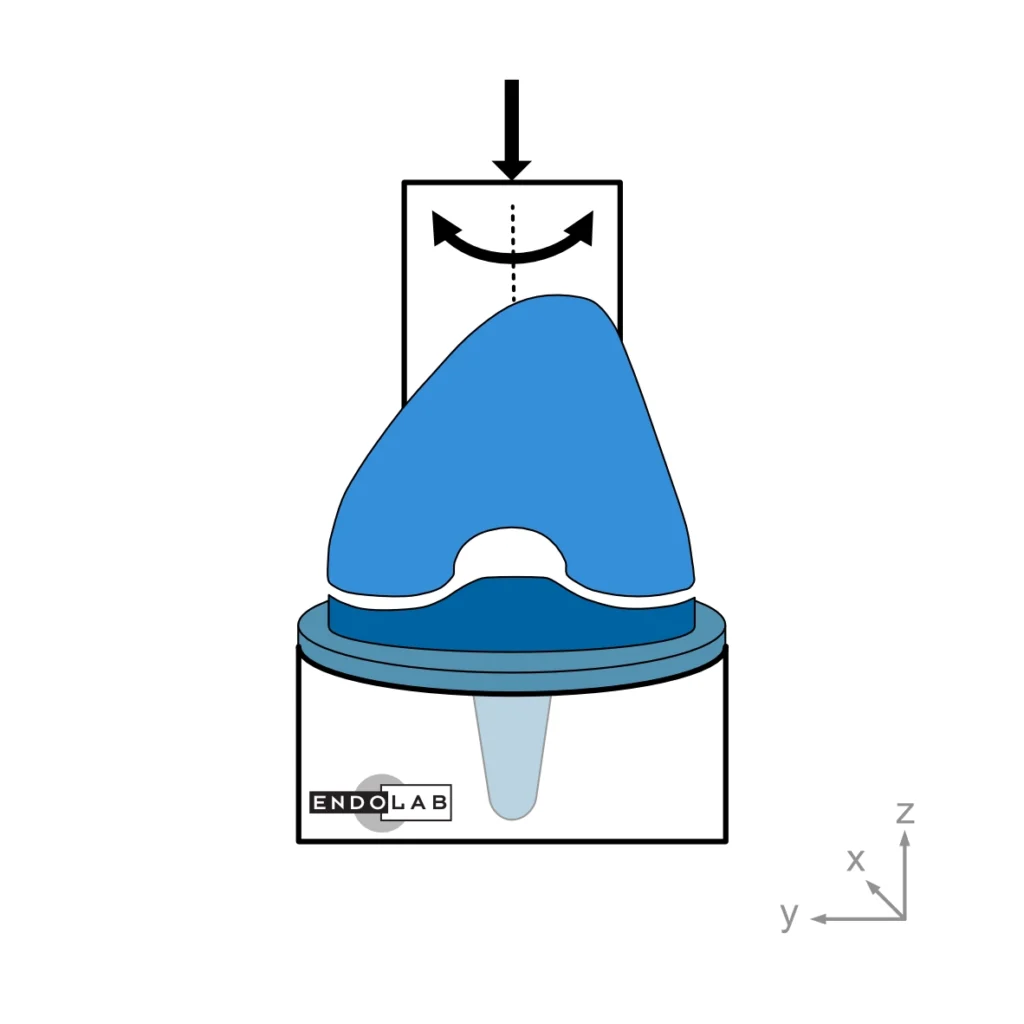
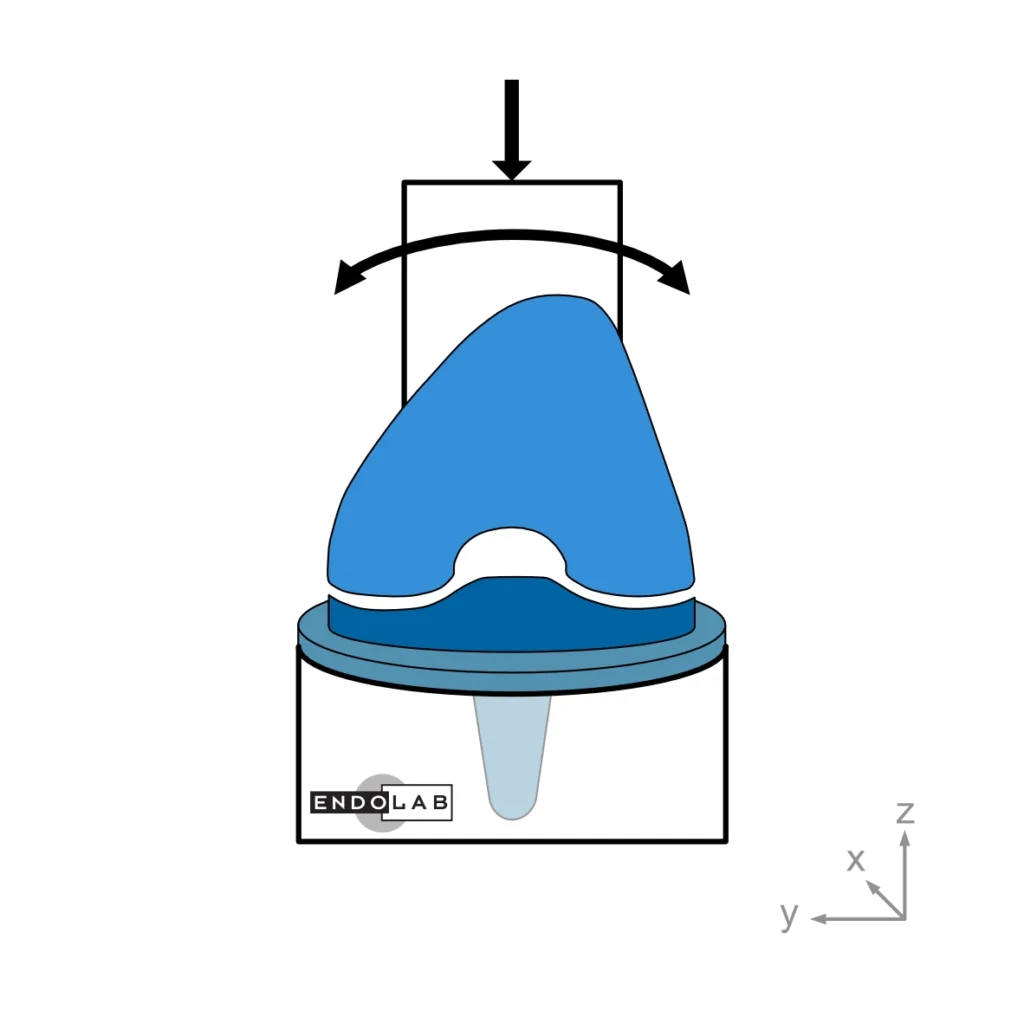
ASTM F1223: Standard Test Method for Determination of Total Knee Replacement Constraint
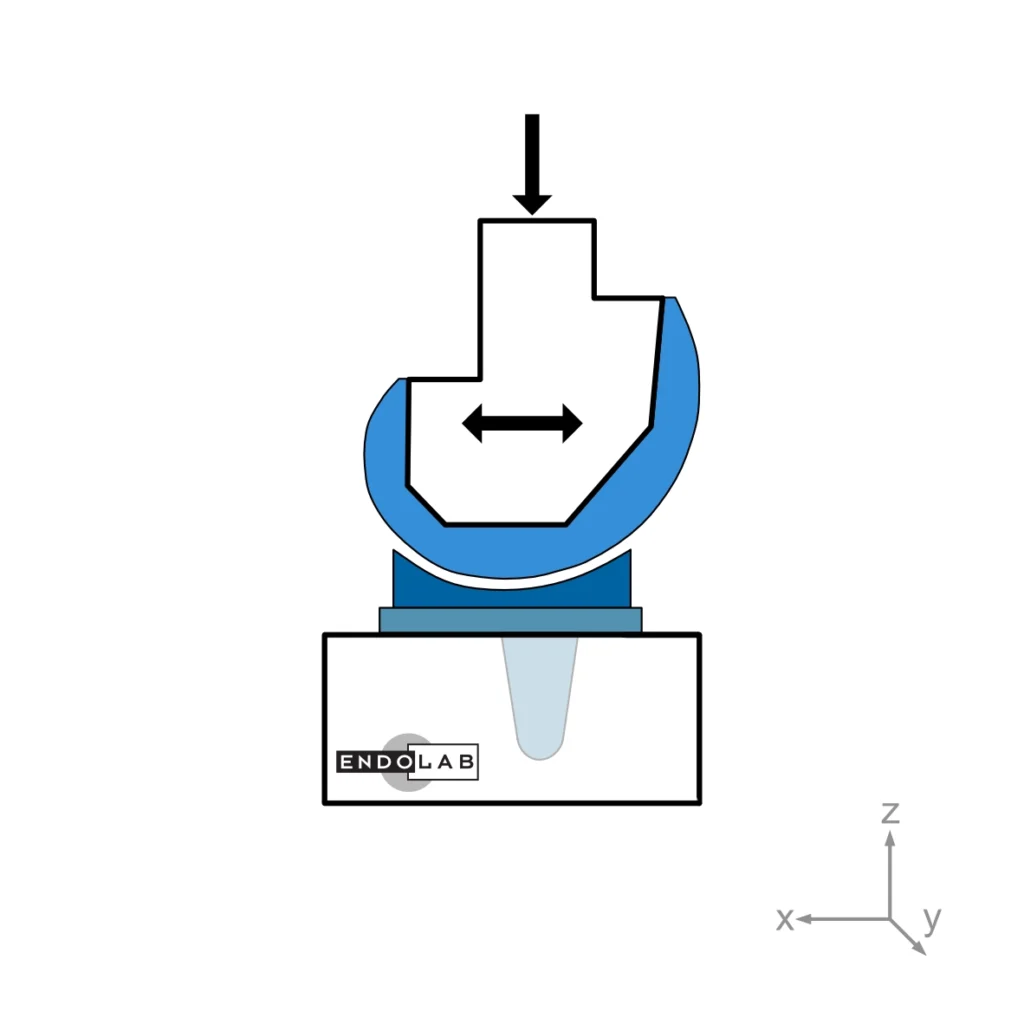
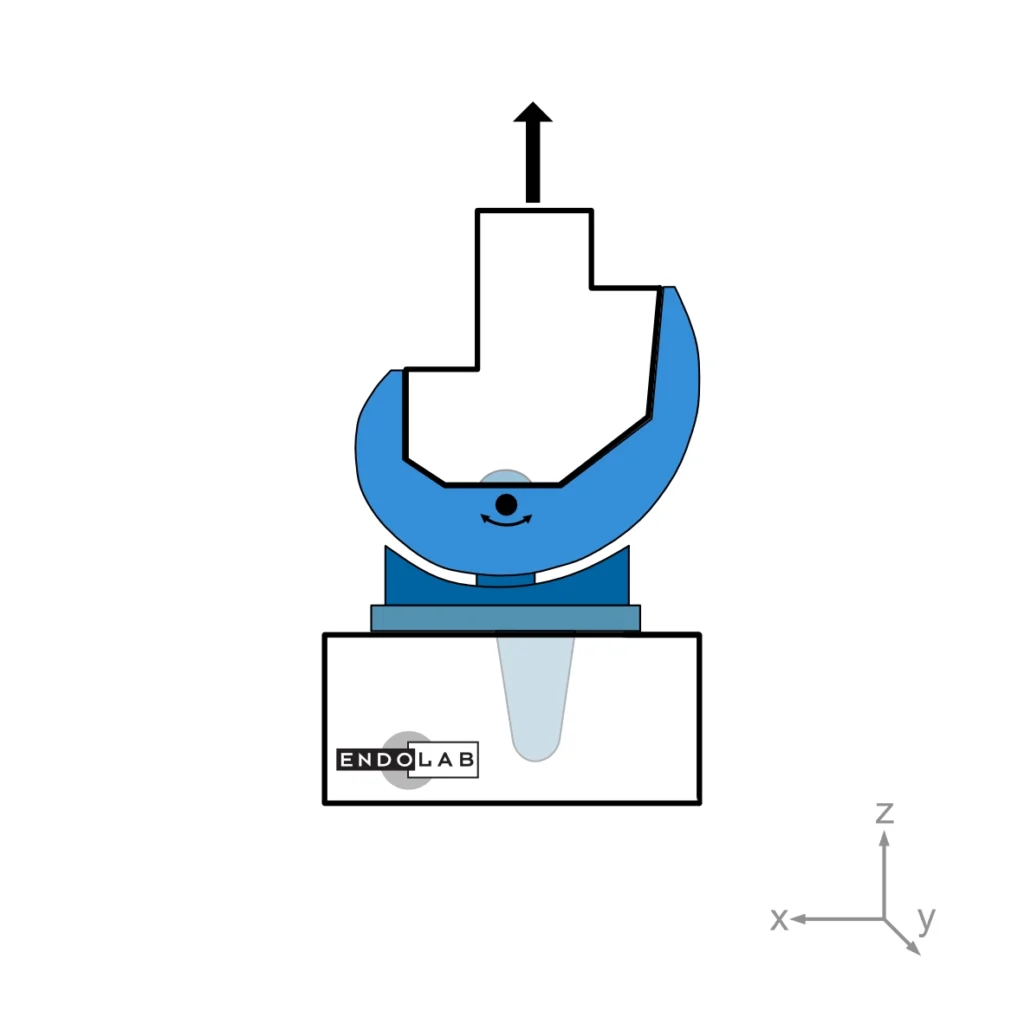
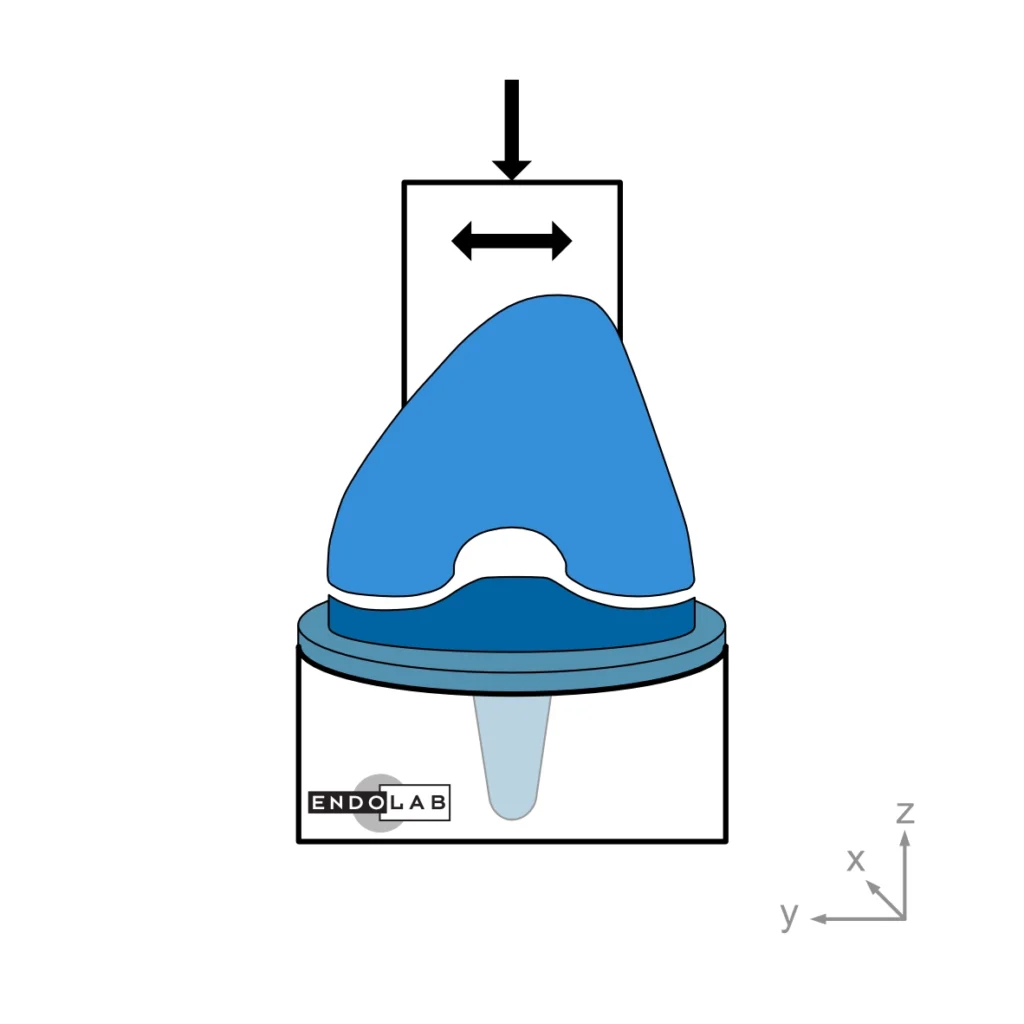
This test method evaluates a total knee replacement’s motion and constraint characteristics subjected to specific loading conditions in an in-vitro environment. More explicitly, it evaluates the ability of the total knee replacement components to withstand anterior-posterior and medial-lateral loads as well as internal-external torques.
In general, the medium (standard) size is tested. The tests are performed at room temperature while keeping the surfaces wet.