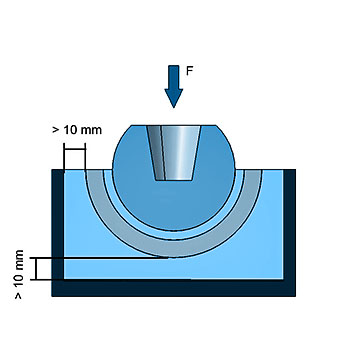
PI-11: Static/Dynamic Compressive Load Test – Hip Insert
This internal test procedure describes the static and dynamic compression testing of modular hip cup inserts. Due to the variety of designs used clinically, modifications to this test procedure may be required for implants with special design features.
The applied loads and loading directions represent a simplification of the in vivo loading conditions. Special consideration shall be given to asymmetric implants and implants with special design features, as these may require modified and/or additional tests.
For the ultimate compression strength test, a static load is applied until specimen failure. For fatigue testing, a dynamic load is applied until either a predefined number of cycles is reached or failure occurs.
After fatigue testing, an ultimate compression strength test may be performed on ceramic inserts to evaluate post-fatigue compression strength. For polymeric inserts, a fixation strength test according to ASTM F1820 may be performed to evaluate post-fatigue fixation performance.